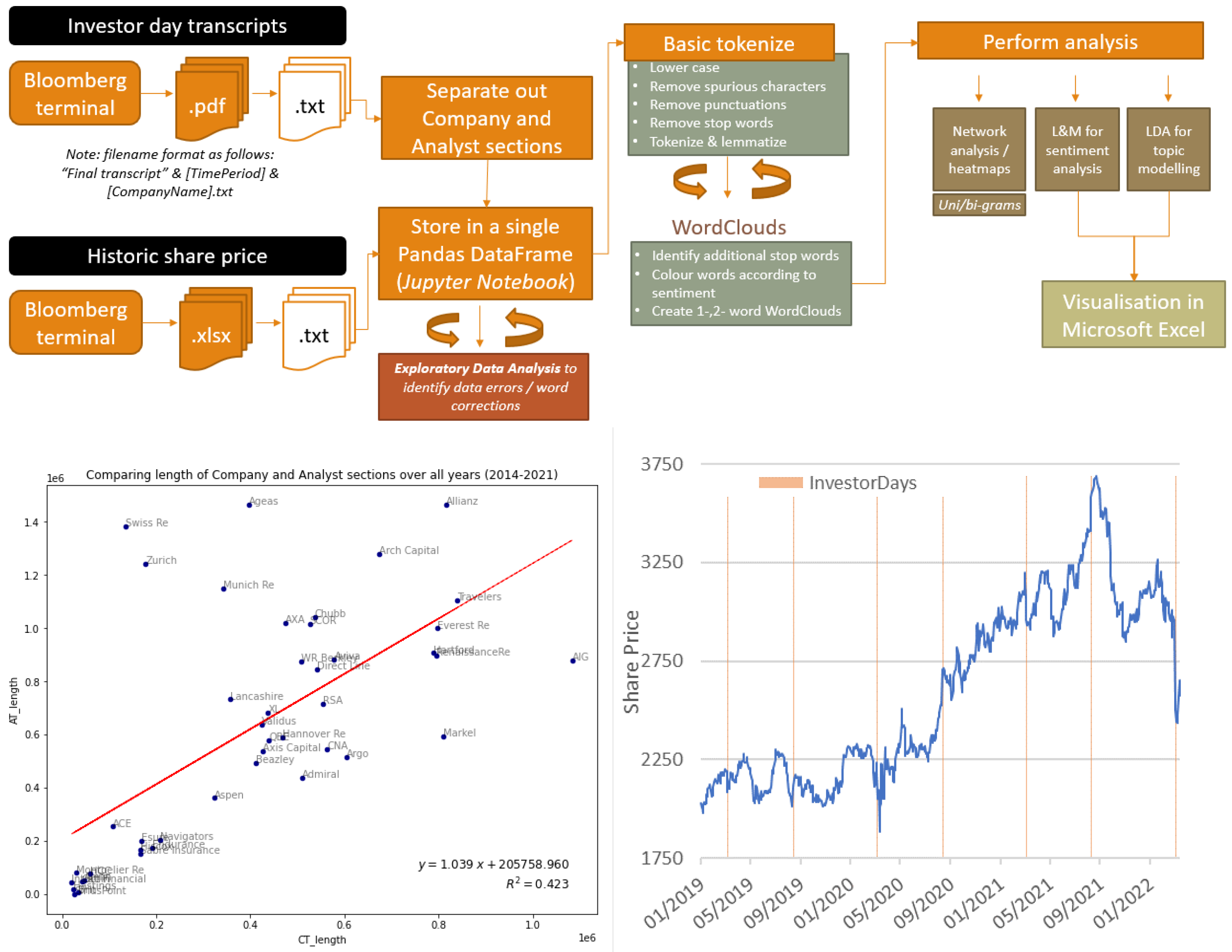
Future Internet | Free Full-Text | Natural Language Processing and Cognitive Networks Identify UK Insurers’ Trends in Investor Day Transcripts

SOLVED: The reduction of tin (IV) oxide via carbon monoxide will produce tin metal and carbon dioxide tnrongh following reaction at 25"C: OVERALL SnOzt) CO,() The stepwise reaction and their 'corresponding equilibrium

Matillion Recognized as a UK Leading Tech Company by Future Fifty; Matillion ETL Named Overall Data Tech Solution of The Year by Data Breakthrough Awards | Matillion

Inner Wolf - Modelling the Hurtta Casual Quilted Overall. http://www.innerwolf.co.uk/hurtta-casual-quilted-overall.html #hurtta #hurttacollection #casual #dogs #dogsofinstagram #dog #overall #onesie #warmth | Facebook
![What is the order with respect to each of the reactant and overall order of the following reactions ? (a) 5Br((aq))^(-)+BrO(3(aq))^(-)+6H((aq))^(+)rarr3Br(2((l)))+3H2O((I)) The experimental rate law is Rate =k[Br^(-)][BrO3^-][H^+]^2 (b) CH3CHO((g ... What is the order with respect to each of the reactant and overall order of the following reactions ? (a) 5Br((aq))^(-)+BrO(3(aq))^(-)+6H((aq))^(+)rarr3Br(2((l)))+3H2O((I)) The experimental rate law is Rate =k[Br^(-)][BrO3^-][H^+]^2 (b) CH3CHO((g ...](https://d10lpgp6xz60nq.cloudfront.net/web-thumb/404708461_web.png)
What is the order with respect to each of the reactant and overall order of the following reactions ? (a) 5Br((aq))^(-)+BrO(3(aq))^(-)+6H((aq))^(+)rarr3Br(2((l)))+3H2O((I)) The experimental rate law is Rate =k[Br^(-)][BrO3^-][H^+]^2 (b) CH3CHO((g ...





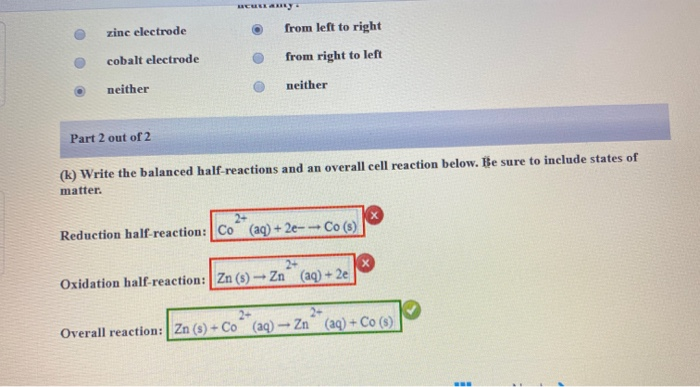

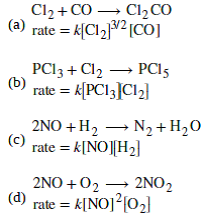

:max_bytes(150000):strip_icc()/CEO-81387466341c465aa8017e70939db730.jpg)
/cloudfront-us-east-1.images.arcpublishing.com/gray/EY3KCTV5IJGODCUUGIXAXCAA3I.jpg)