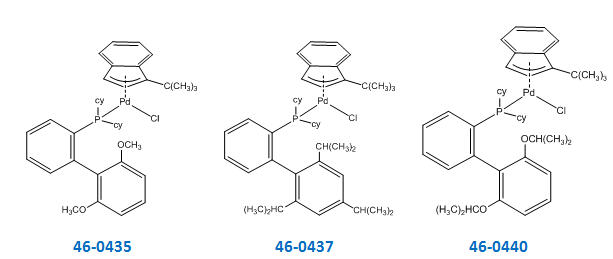
Palladium-Catalyzed C–P Bond-Forming Reactions of Aryl Nonaflates Accelerated by Iodide | The Journal of Organic Chemistry

Scheme 1 A catalytic cycle for palladium(0)-mediated benzylphosphonate... | Download Scientific Diagram

Homogeneous Palladium Catalyst Suppressing Pd Black Formation in Air Oxidation of Alcohols | Journal of the American Chemical Society

Formation of Palladium(0) Complexes from Pd(OAc)2 and a Bidentate Phosphine Ligand (dppp) and Their Reactivity in Oxidative Addition | Organometallics
Synthesis of dipalladium(III) complexes with Pd-Pd bonds. Formation... | Download Scientific Diagram

Palladium-catalyzed microwave-assisted Hirao reaction utilizing the excess of the diarylphosphine oxide reagent as the P-ligand; a study on the activity and formation of the “PdP2” catalyst

Formation of Palladium(0) Complexes from Pd(OAc)2 and a Bidentate Phosphine Ligand (dppp) and Their Reactivity in Oxidative Addition | Organometallics

PPT – Development of Palladium-Catalyzed C-N Bond Formation Reaction PowerPoint presentation | free to view - id: 3d4352-YWY3N

Palladium‐Catalyzed C–S Bond Formation: Rate and Mechanism of the Coupling of Aryl or Vinyl Halides with a Thiol Derived from a Cysteine - Moreau - 2005 - European Journal of Organic Chemistry -

Formation and propagation of well-defined Pd nanoparticles (PdNPs) during C–H bond functionalization of heteroarenes: are nanoparticles a moribund form of Pd or an active catalytic species? - ScienceDirect

Palladium-Catalyzed β-Elimination of Aminoboranes from (Aminomethylsilyl)boranes Leading to the Formation of Silene Dimers | Organometallics

PDF) Palladium-mediated intramolecular C–N bond formation involving allyl substituted pyridines. Application to a novel strategy for the synthesis of the skeleton of berberinium derivatives | Mike Robitzer, Michel Pfeffer, and Gerard Van

Palladium-Catalyzed Direct Intramolecular C-N Bond Formation: Access to Multisubstituted Dihydropyrroles

Formation of Palladium(0) Complexes from Pd(OAc)2 and a Bidentate Phosphine Ligand (dppp) and Their Reactivity in Oxidative Addition | Organometallics

Formation of Enamides via Palladium(II)-Catalyzed Vinyl Transfer from Vinyl Ethers to Nitrogen Nucleophiles | Organic Letters

Formation of palladium complexes of ligands 2a, 5 and 7. (A) Formation... | Download Scientific Diagram
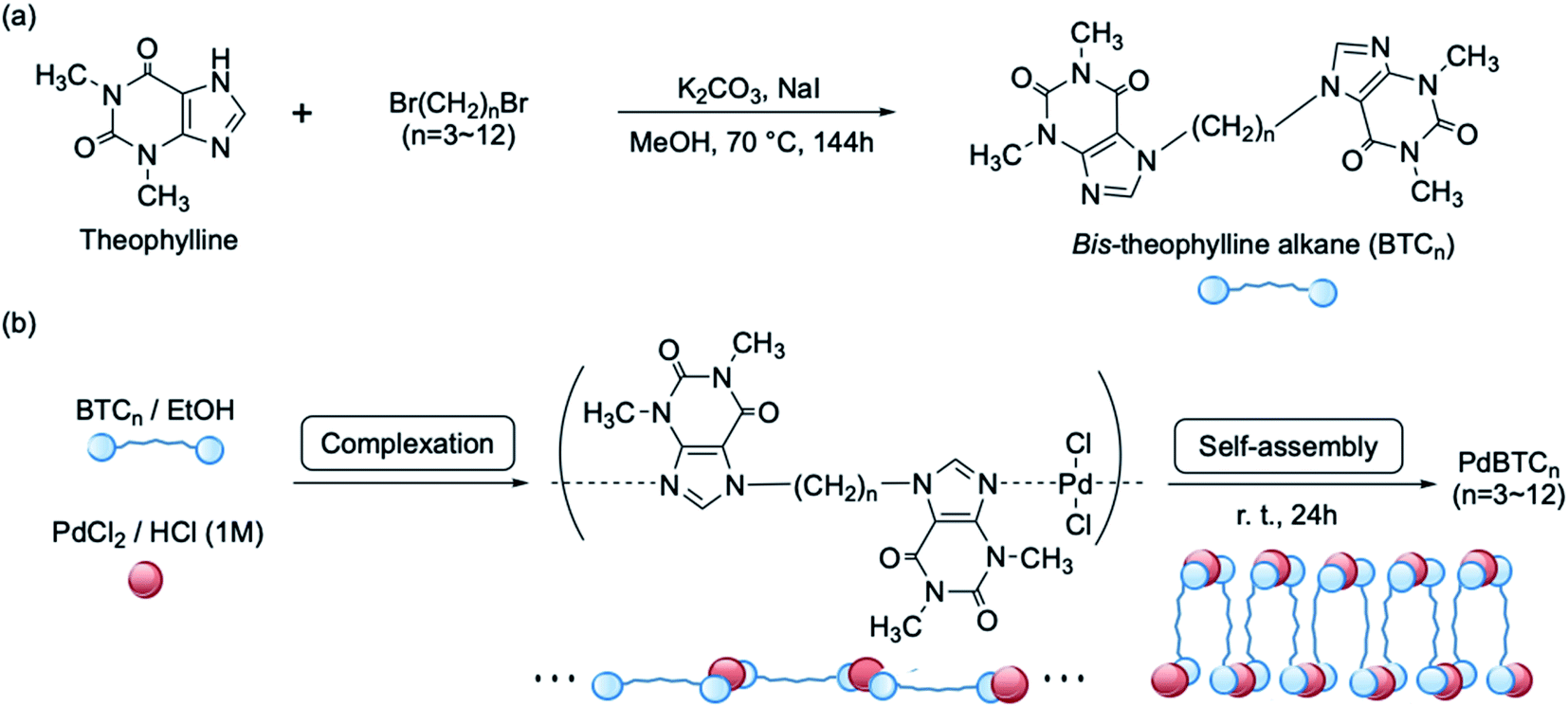
Microflowers formed by complexation-driven self-assembly between palladium( ii ) and bis-theophyllines: immortal catalyst for C–C cross-coupling react ... - RSC Advances (RSC Publishing) DOI:10.1039/D1RA06177A
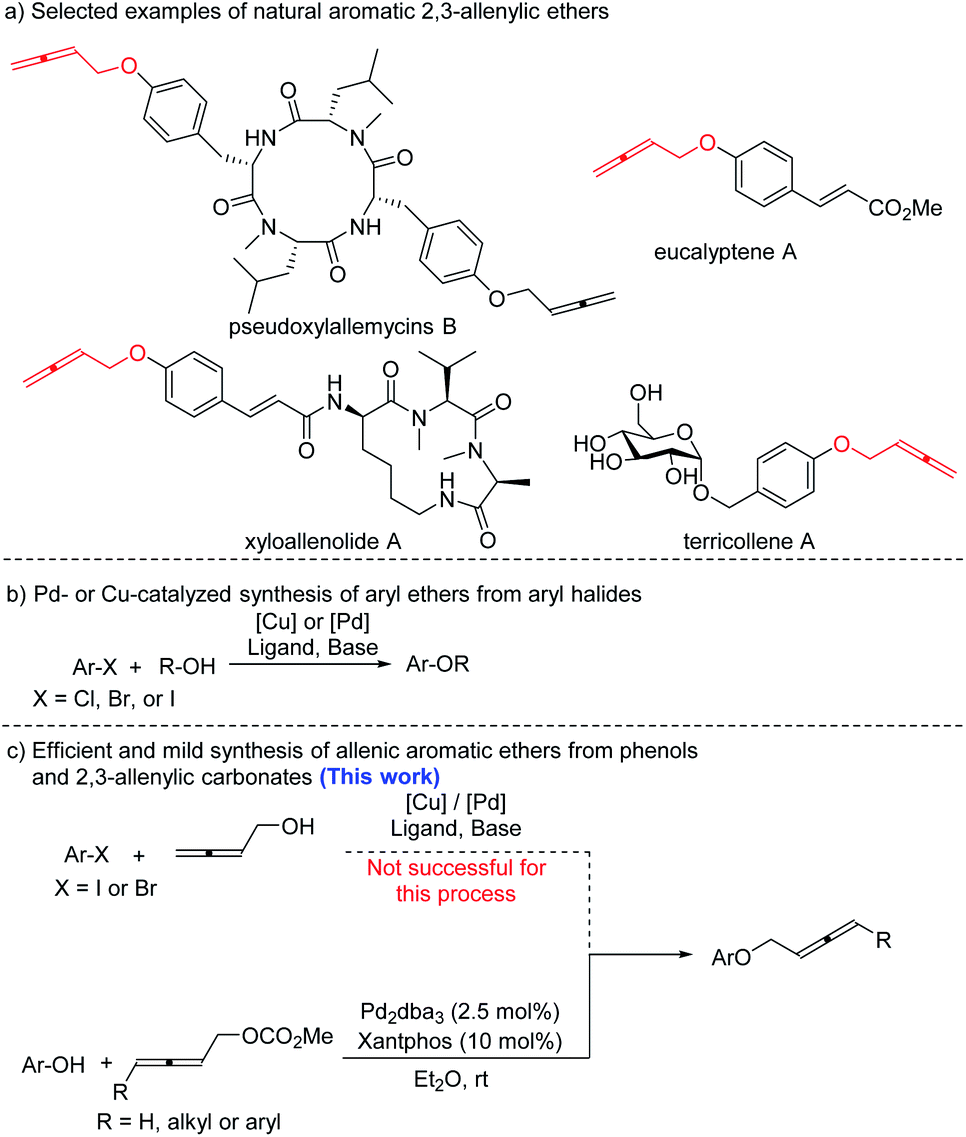
A palladium-catalyzed approach to allenic aromatic ethers and first total synthesis of terricollene A - Chemical Science (RSC Publishing) DOI:10.1039/D1SC01896E

Palladium-catalyzed C–P(III) bond formation reaction with acylphosphines as phosphorus source - ScienceDirect