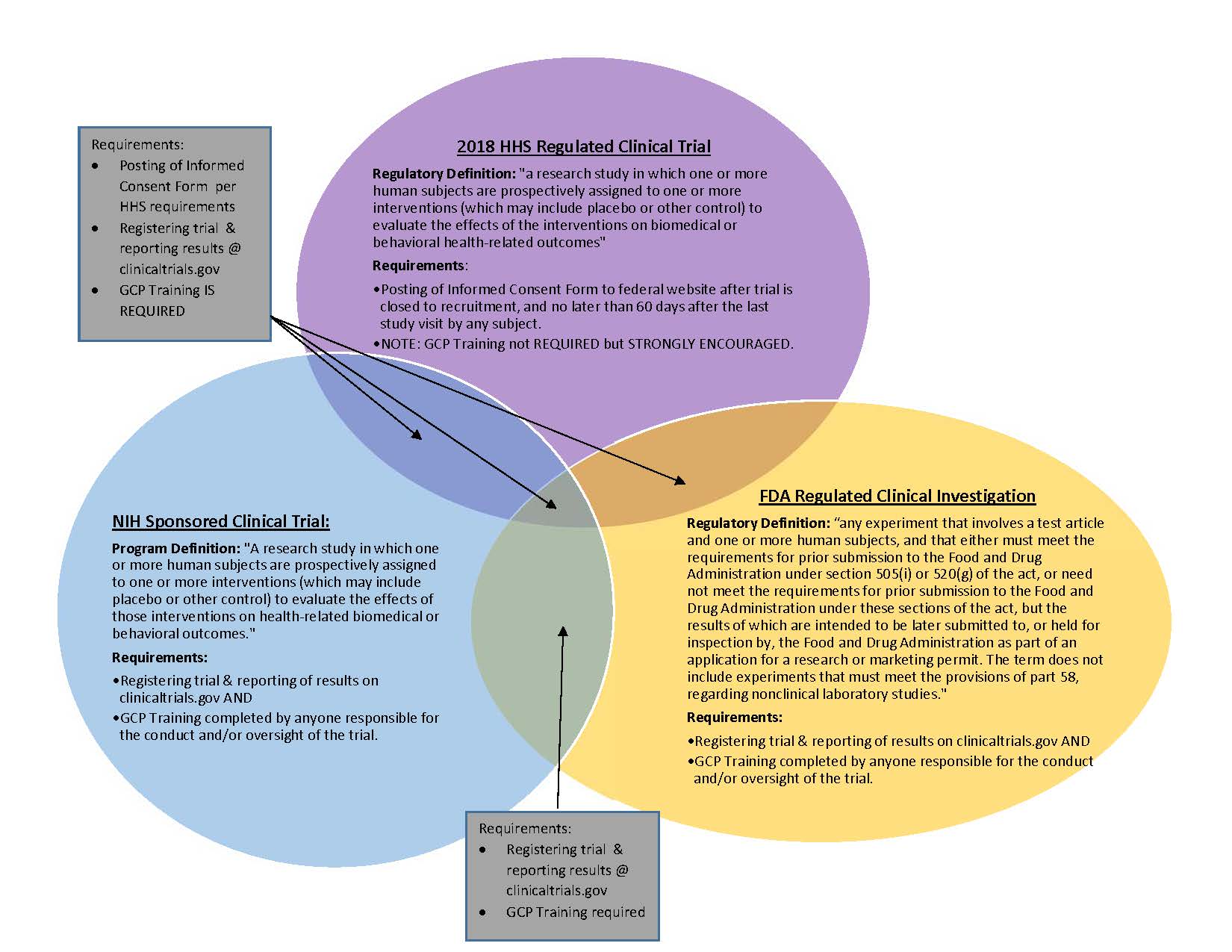
Design characteristics, risk of bias, and reporting of randomised controlled trials supporting approvals of cancer drugs by European Medicines Agency, 2014-16: cross sectional analysis | The BMJ

Science disconnected: the translational gap between basic science, clinical trials, and patient care in Alzheimer's disease - The Lancet Healthy Longevity
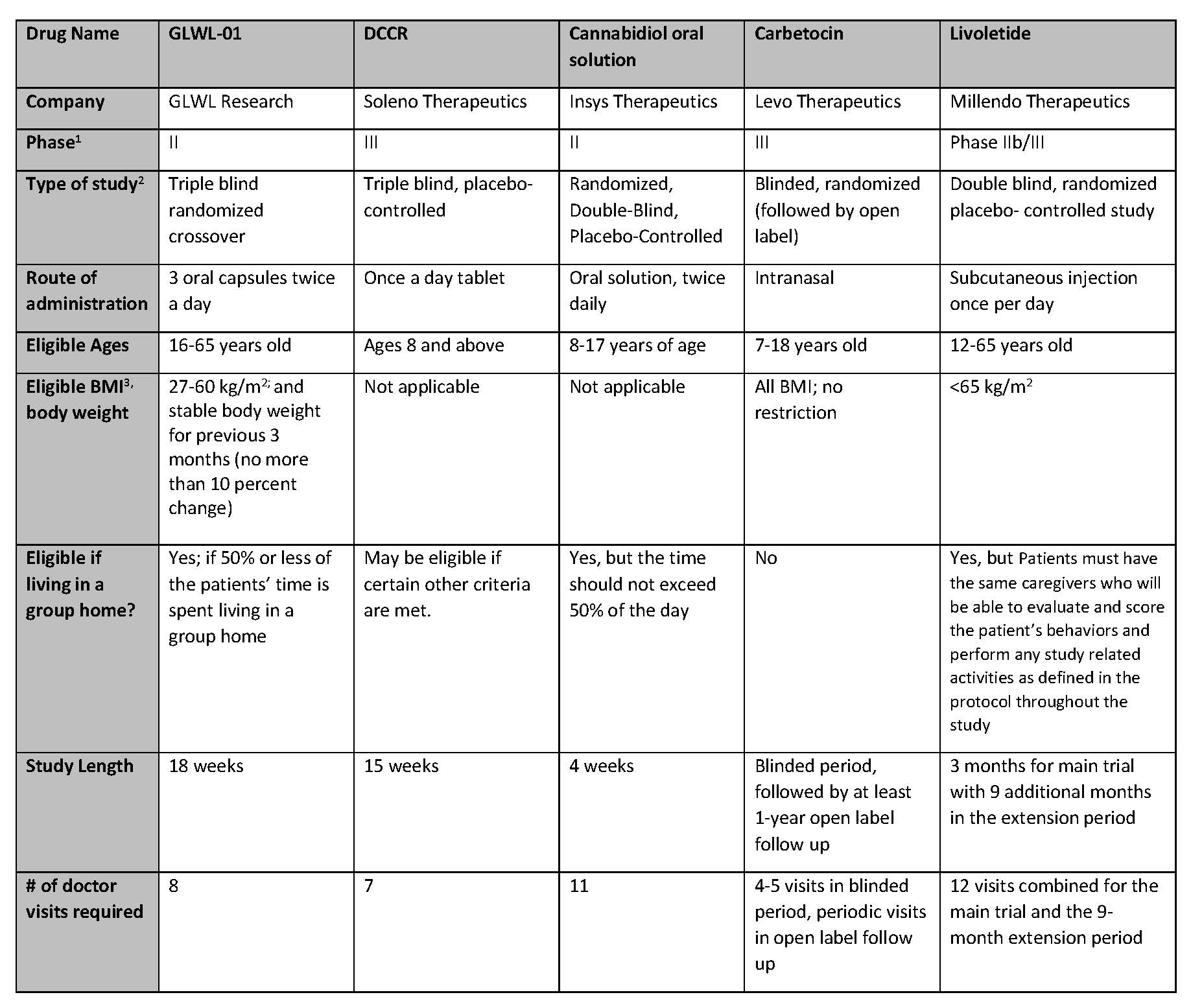
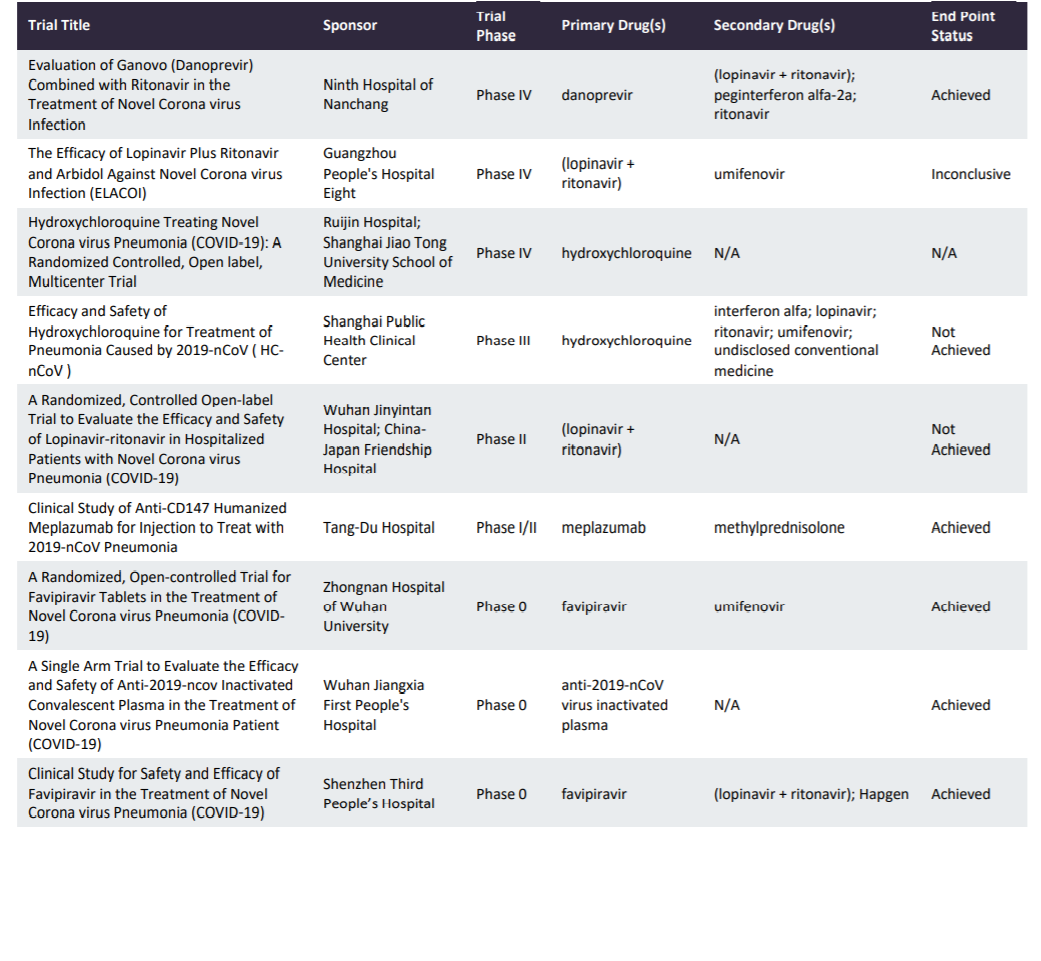
Summary Of Active Clinical Trials For Prader-Willi Syndrome Hyperphagia - Prader-Willi Syndrome Association | USA
Evidence for the Selective Reporting of Analyses and Discrepancies in Clinical Trials: A Systematic Review of Cohort Studies of Clinical Trials | PLOS Medicine

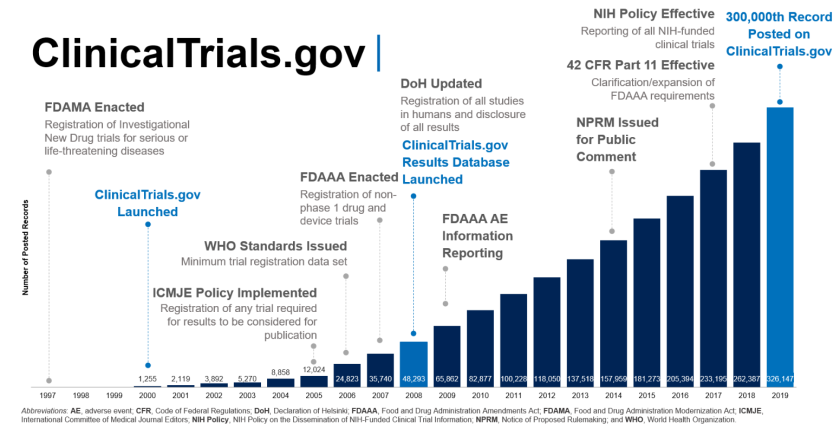
Compliance with legal requirement to report clinical trial results on ClinicalTrials.gov: a cohort study - The Lancet

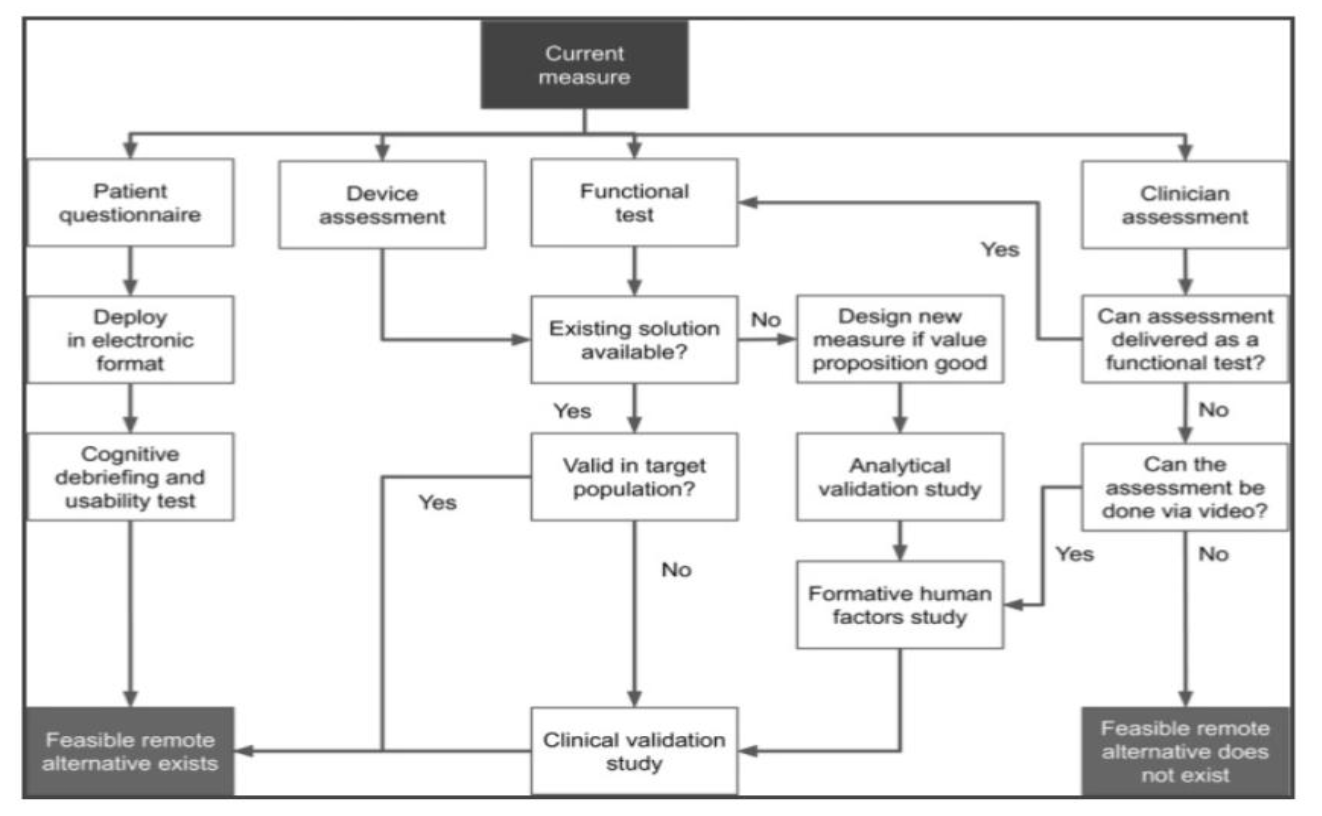
Clinical trials site recruitment optimisation: Guidance from Clinical Trials: Impact and Quality - Christine Zahren, Sonia Harvey, Leanne Weekes, Charlotte Bradshaw, Radhika Butala, John Andrews, Sally O'Callaghan, 2021