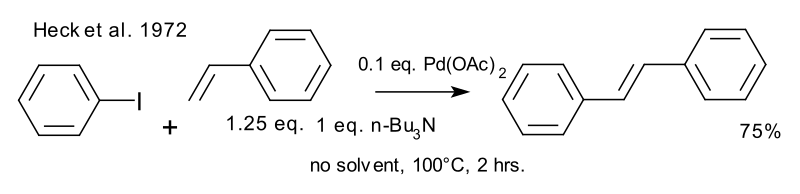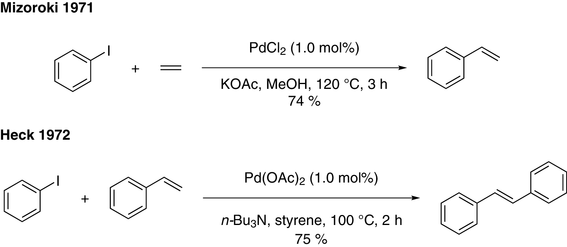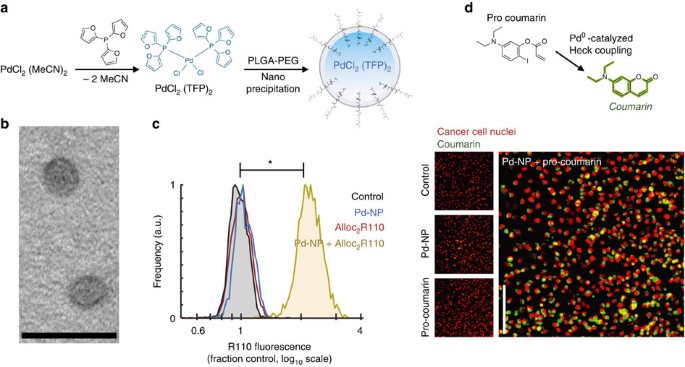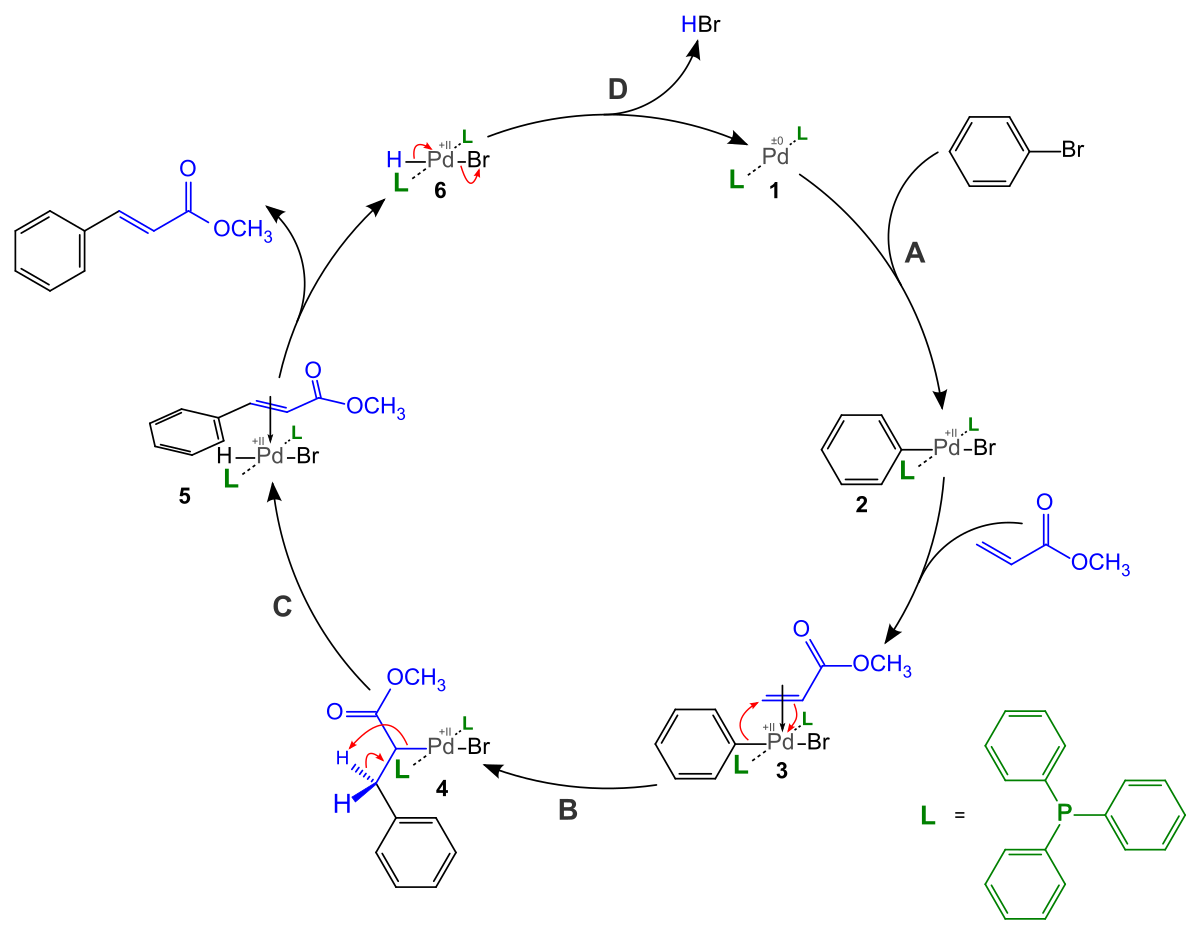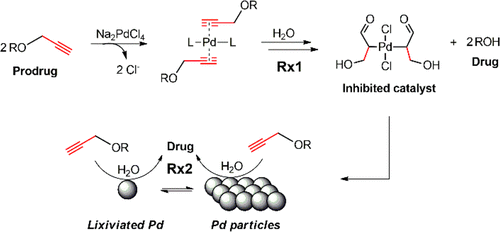
Mechanism of Palladium(II)-Mediated Uncaging Reactions of Propargylic Substrates,ACS Catalysis - X-MOL

Molecules | Free Full-Text | Selective Heck Arylation of Cyclohexene with Homogeneous and Heterogeneous Palladium Catalysts | HTML

Recent Developments in Metal‐Catalyzed Bio‐orthogonal Reactions for Biomolecule Tagging - Jang - 2019 - ChemBioChem - Wiley Online Library
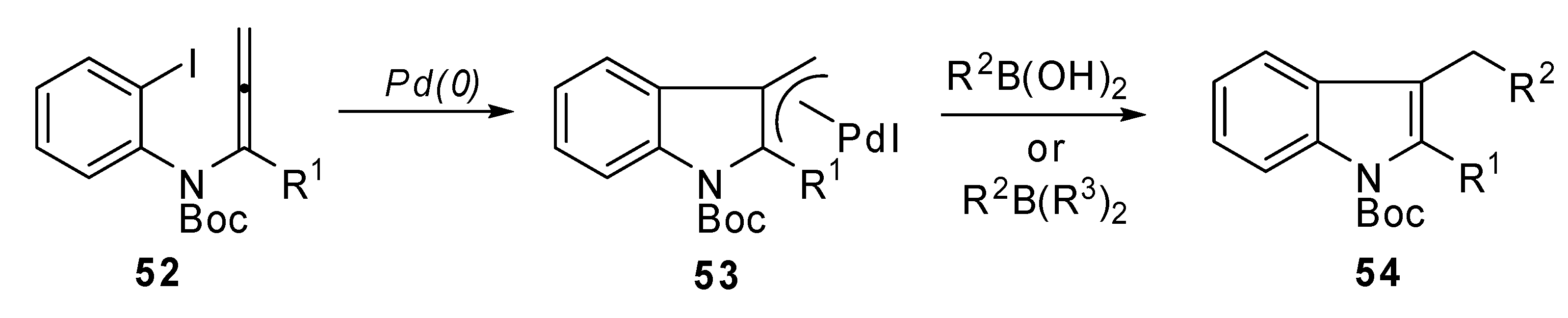
Molecules | Free Full-Text | The Heck Reaction Applied to 1,3- and 1,2-Unsaturated Derivatives, a Way towards Molecular Complexity | HTML
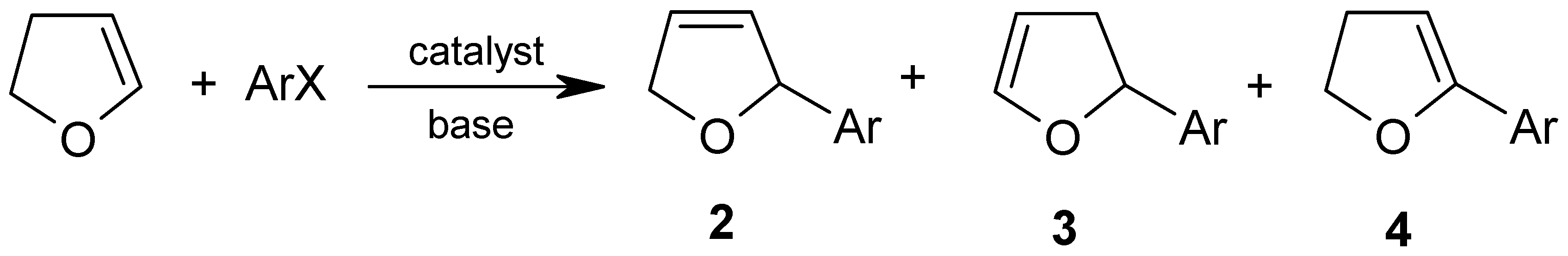
Molecules | Free Full-Text | Palladium Catalyzed Heck Arylation of 2,3-Dihydrofuran—Effect of the Palladium Precursor | HTML

Molecules | Free Full-Text | The Heck Reaction Applied to 1,3- and 1,2-Unsaturated Derivatives, a Way towards Molecular Complexity | HTML
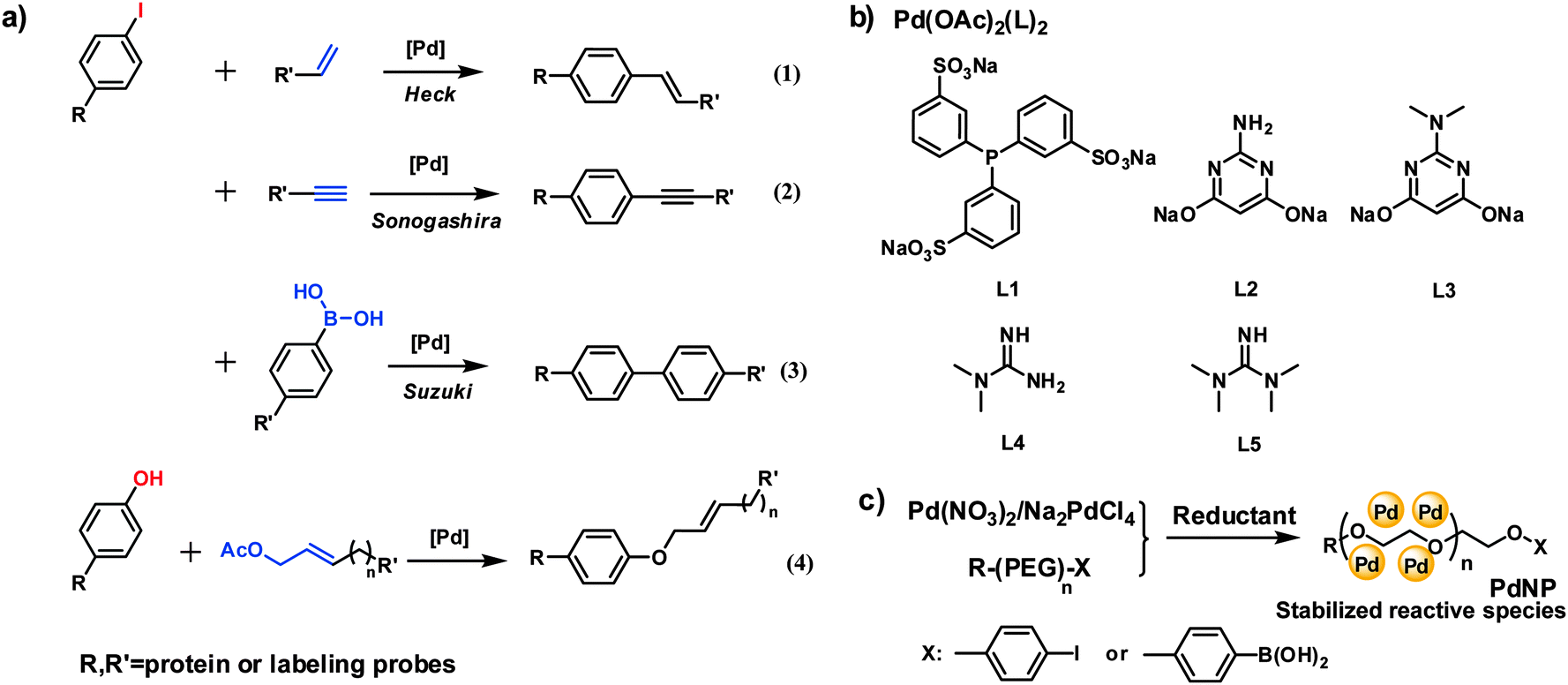
Transition metal-mediated bioorthogonal protein chemistry in living cells - Chemical Society Reviews (RSC Publishing)
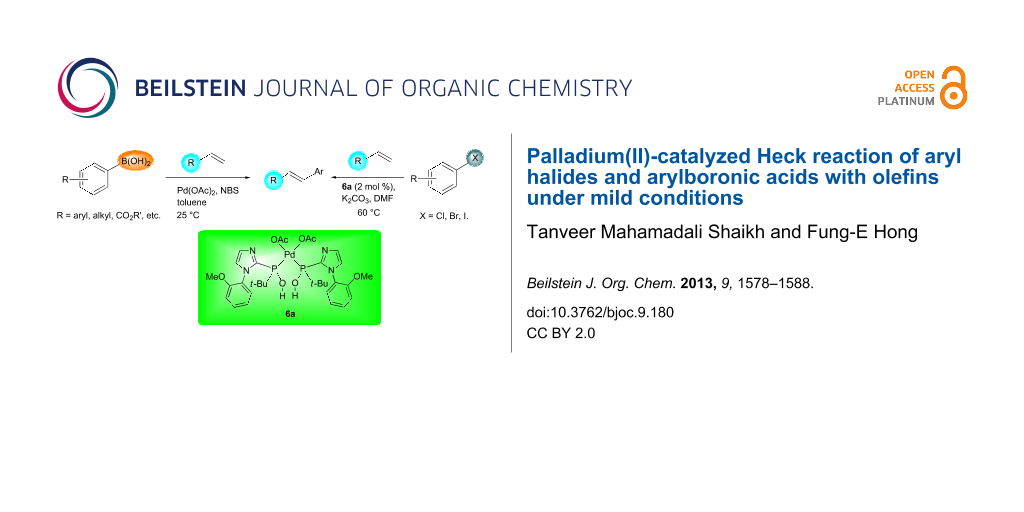
Palladium(II)-catalyzed Heck reaction of aryl halides and arylboronic acids with olefins under mild conditions
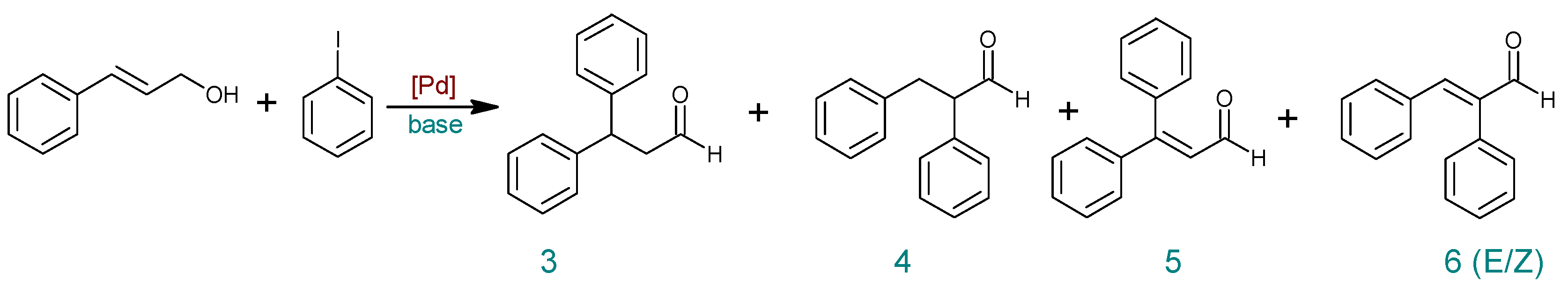
Molecules | Free Full-Text | Heck Transformations of Biological Compounds Catalyzed by Phosphine-Free Palladium | HTML
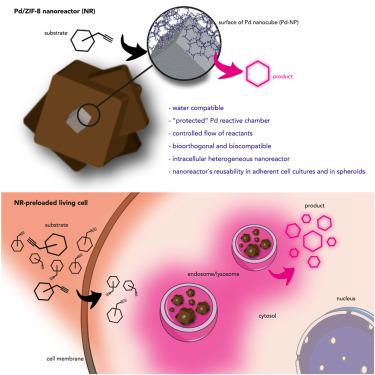
Core-Shell Palladium/MOF Platforms as Diffusion-Controlled Nanoreactors in Living Cells and Tissue Models. - Cell Rep. Phys. Sci. - X-MOL
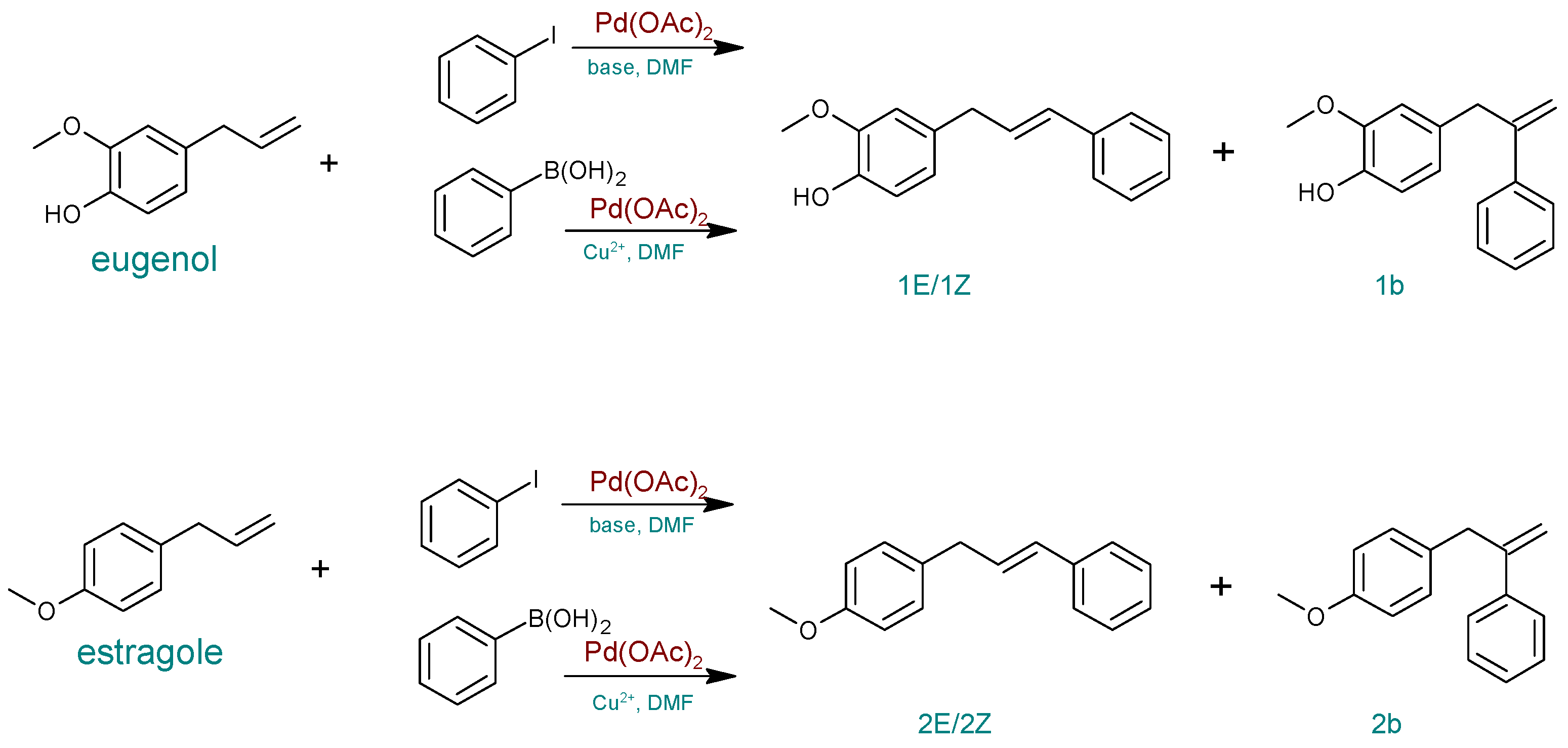
Molecules | Free Full-Text | Heck Transformations of Biological Compounds Catalyzed by Phosphine-Free Palladium | HTML

Core-Shell Palladium/MOF Platforms as Diffusion-Controlled Nanoreactors in Living Cells and Tissue Models - ScienceDirect
Catalytic modification of dehydroalanine in peptides and proteins via palladium mediated cross coupling