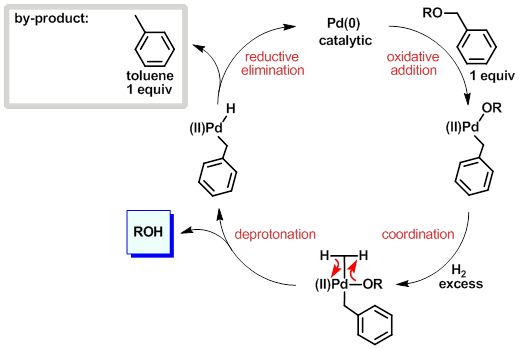
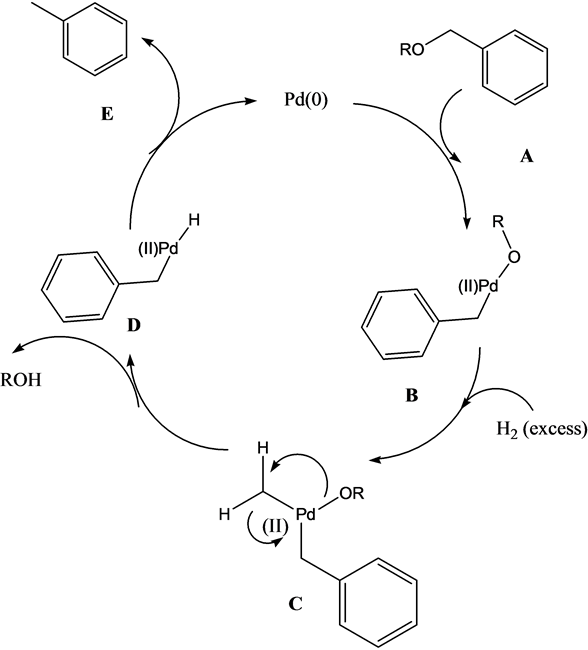Facile and selective cleavage of allyl ethers, amines and esters using polymethylhydrosiloxane–ZnCl2/Pd(PPh3)4 - ScienceDirect

Palladium-catalyzed reaction of tributyltin hydride. Selective and very mild deprotection of allyl and allyloxycarbonyl derivatives of amino-acids
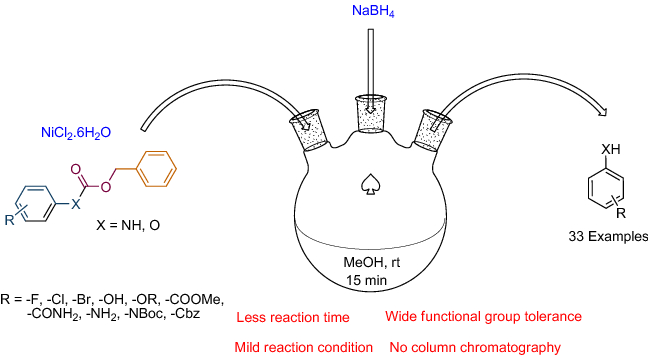
Development of a novel protocol for chemoselective deprotection of N / O -benzyloxycarbonyl (Cbz) at ambient temperature | SpringerLink

Optimization of reaction conditions for deprotection along with cross... | Download Scientific Diagram

PDF) A concise Pd catalyzed cross coupling reaction along with deprotection for the synthesis of a new series of pyrimidine derivatives
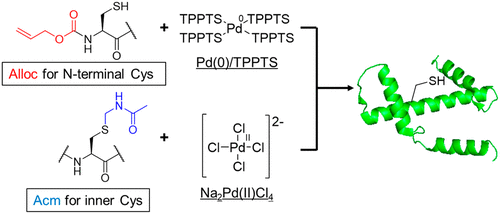
Chemical Synthesis of Cys-Containing Protein via Chemoselective Deprotection with Different Palladium Complexes.,Organic Letters - X-MOL

Palladium-Catalyzed α-Arylation of Silylenol Ethers in the Synthesis of Isoquinolines and Phenanthridines - Org. Lett. - X-MOL
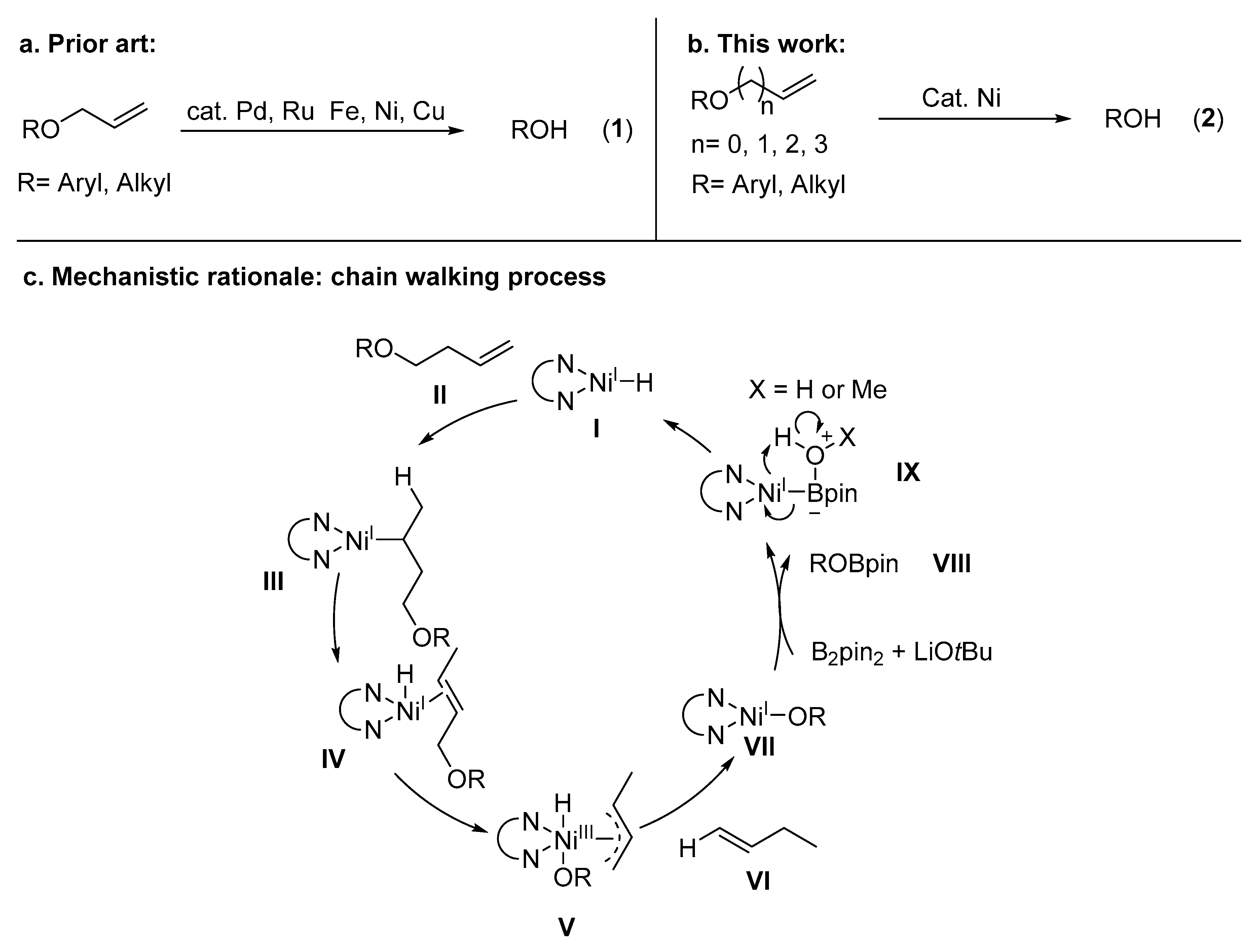
Molecules | Free Full-Text | Nickel-Catalyzed Removal of Alkene Protecting Group of Phenols, Alcohols via Chain Walking Process | HTML

Allylic protecting groups and their use in a complex environment part II: Allylic protecting groups and their removal through catalytic palladium π-allyl methodology
![PDF] Palladium-triggered deprotection chemistry for protein activation in living cells. | Semantic Scholar PDF] Palladium-triggered deprotection chemistry for protein activation in living cells. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3ae70a978b403098bfa58487a5f2c84d3e816087/2-Figure1-1.png)
PDF] Palladium-triggered deprotection chemistry for protein activation in living cells. | Semantic Scholar

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds
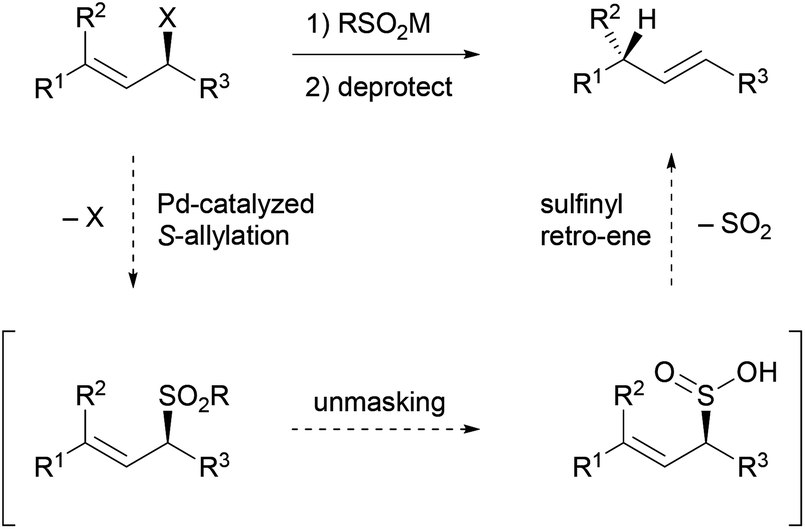
Stereoselective allylic reduction via one-pot palladium-catalyzed allylic sulfonation and sulfinyl retro-ene reactions - Organic Chemistry Frontiers (RSC Publishing)

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds

Palladium-triggered deprotection chemistry for protein activation in living cells | Nature Chemistry
A Mechanistic Study of Direct Activation of Allylic Alcohols in Palladium Catalyzed Amination Reactions

Intracellular Deprotection Reactions Mediated by Palladium Complexes Equipped with Designed Phosphine Ligands. - Abstract - Europe PMC

Intracellular Deprotection Reactions Mediated by Palladium Complexes Equipped with Designed Phosphine Ligands. - Abstract - Europe PMC