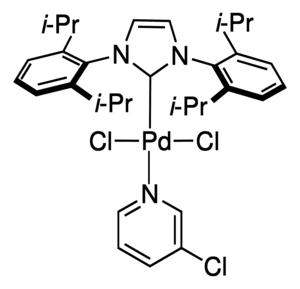
Cycloheptyl substituted N-heterocyclic carbene PEPPSI-type palladium complexes with different N-coordinated ligands: Involvement in Suzuki-Miyaura reaction - ScienceDirect
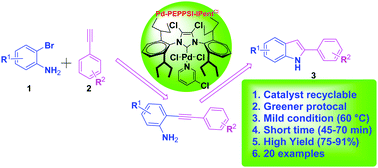
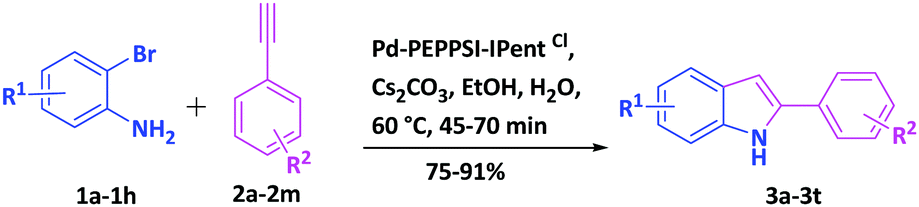
Pd-PEPPSI-IPentCl: a new highly efficient ligand-free and recyclable catalyst system for the synthesis of 2-substituted indoles via domino copper-free Sonogashira coupling/cyclization - New Journal of Chemistry (RSC Publishing)

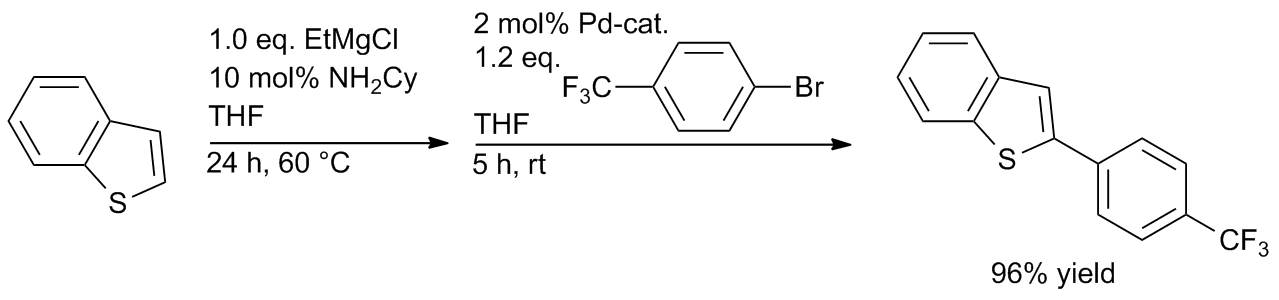
Pd‐PEPPSI: Water‐Assisted Suzuki−Miyaura Cross‐Coupling of Aryl Esters at Room Temperature using a Practical Palladium‐NHC (NHC=N‐Heterocyclic Carbene) Precatalyst - Li - 2018 - Advanced Synthesis & Catalysis - Wiley Online Library

Understanding existing and designing novel synthetic routes to Pd-PEPPSI-NHC and Pd-PEPPSI-PR 3 pre-catalysts - Chemical Communications (RSC Publishing) DOI:10.1039/D0CC02262D
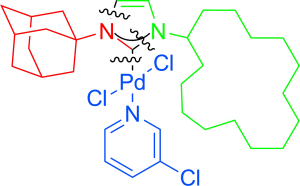
An Alternative Approach to PEPPSI Catalysts: From Palladium Isonitriles to Highly Active Unsymmetrically Substituted PEPPSI Catalysts - Chem. Eur. J. - X-MOL
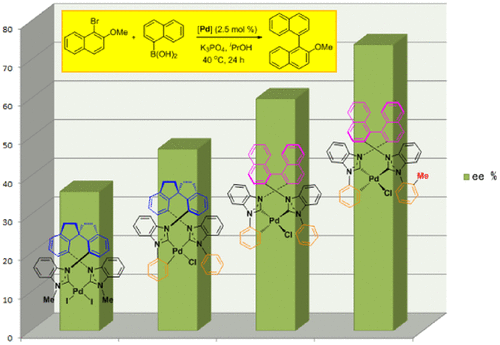
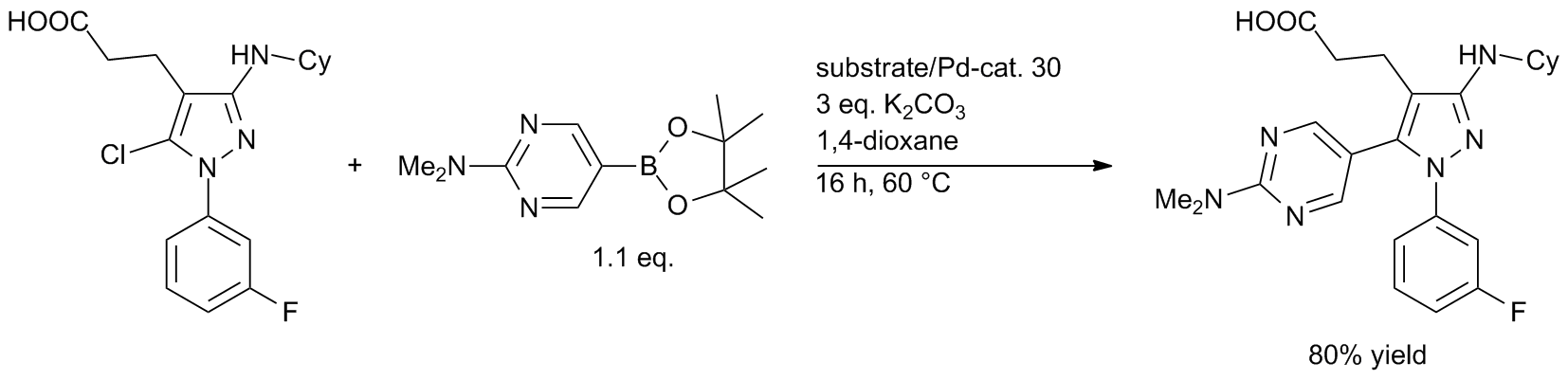
Fine Tuning of Chiral Bis(N-heterocyclic carbene) Palladium Catalysts for Asymmetric Suzuki–Miyaura Cross-Coupling Reactions: Exploring the Ligand Modification,Organometallics - X-MOL
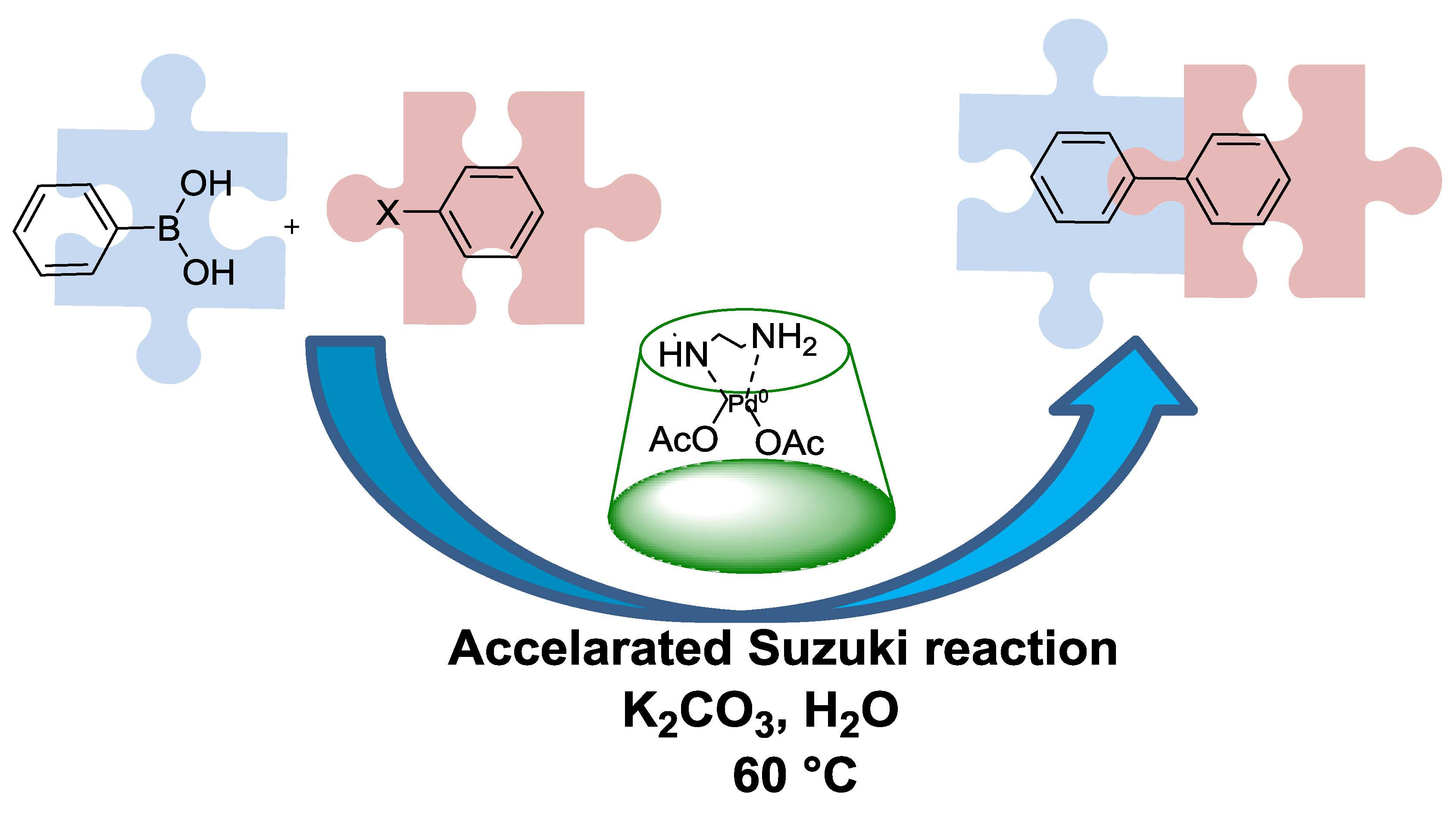
Catalysts | Free Full-Text | An Amino-Chain Modified β-cyclodextrin: A Supramolecular Ligand for Pd(OAc)2 Acceleration in Suzuki–Miyaura Coupling Reactions in Water
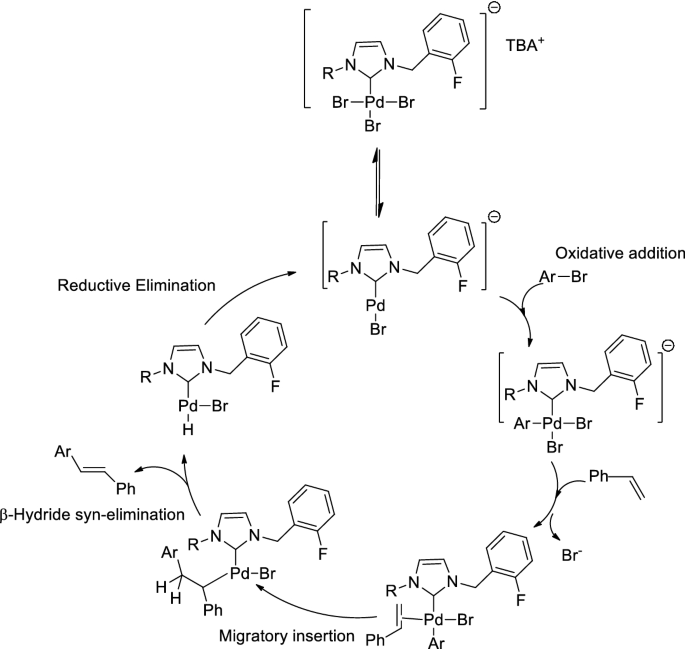
A new PEPPSI type N-heterocyclic carbene palladium(II) complex and its efficiency as a catalyst for Mizoroki-Heck cross-coupling reactions in water | SpringerLink

Figure 1 from PEPPSI-type palladium complexes containing basic 1,2,3-triazolylidene ligands and their role in Suzuki-Miyaura catalysis. | Semantic Scholar

PEPPSI‐Type Palladium–NHC Complexes: Synthesis, Characterization, and Catalytic Activity in the Direct C5‐Arylation of 2‐Substituted Thiophene Derivatives with Aryl Halides - Kaloğlu - 2017 - European Journal of Inorganic Chemistry - Wiley Online Library

Catalysts | Free Full-Text | N-Acylphthalimides: Efficient Acyl Coupling Reagents in Suzuki–Miyaura Cross-Coupling by N–C Cleavage Catalyzed by Pd– PEPPSI Precatalysts | HTML
![Various [NHC-Pd-PEPPSI] (left) and long alkyl chain-labeled... | Download Scientific Diagram Various [NHC-Pd-PEPPSI] (left) and long alkyl chain-labeled... | Download Scientific Diagram](https://www.researchgate.net/profile/Muhammad_Ghufran_Rafique/publication/317660984/figure/fig1/AS:528262394638337@1502958977918/Various-NHC-Pd-PEPPSI-left-and-long-alkyl-chain-labeled-NHC-Pdallyl-cinnamyl.png)